
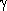 -crystallin B chain
-crystallin B chain
pdb-file for RasMol with script, or load directly pdb-file and script.
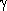 -crystallin D chain
-crystallin D chain
pdb-file for RasMol with script, or load directly pdb-file and script.
Note that for automatic firing up of the display it is a requirement that RasMol is installed and the browser configured (you will learn in the course how to do this).
The biggest sequence difference is a deletion of 1 residue (residue 86 in red) in the
linker connecting the two domains in
(at the bottom of the molecule in the display)
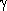 D Crystallin
compared to
D Crystallin
compared to 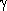 B Crystallin. A structural
difference is that the C-terminal tails have a different conformation. Yet,
it is clear that the three-dimensional structures of both proteins are
at first sight indistinguishable. Proteins have similar three-dimensional
conformations for sequence identities as low as 20%. This phenomenon
is at the basis of molecular replacement.
B Crystallin. A structural
difference is that the C-terminal tails have a different conformation. Yet,
it is clear that the three-dimensional structures of both proteins are
at first sight indistinguishable. Proteins have similar three-dimensional
conformations for sequence identities as low as 20%. This phenomenon
is at the basis of molecular replacement.
The principle of molecular replacement
When identical or similar structures exist in different crystallographic environments, similarities between their diffraction patterns, which are direcly related to their Fourier transforms, would be expected. The technique of Molecular Replacement exploits this similarity to determine phases. The dominant application is
- Different crystal forms of the same or similar molecules (exact, crystallographic
symmetry)
- Similarity or identity of structure in different parts of the crystallographic
asymmetric unit caused by a repetition of the same subunit structure in the
formation of a whole molecule (inexact, local or non-crystallographic symmetry)
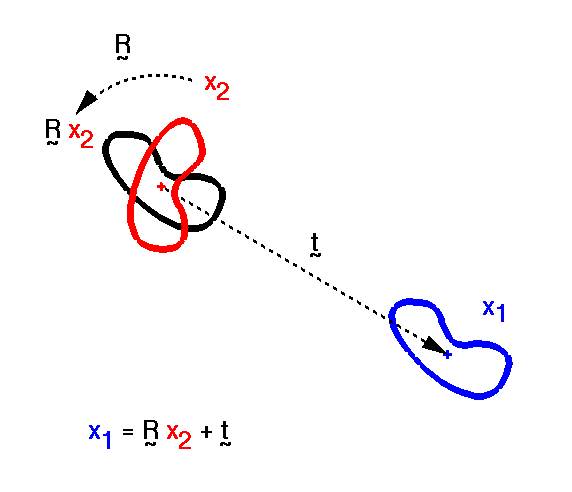
To express the relationship between 2 identical rigid bodies, 6 parameters, 3 rotational and 3 translational, are required.The rigid bodies are described by the coordinates:
x1 unknown, observed or target
x2 known or model
The relationships are described by the operations:
R rotation matrix expressed in 3 Eulerian angles
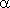
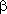
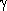 or
or  1
1  2
2
 3
3
t translation vector tx ty tz
Molecular replacement is a six-dimensional problem with order 6, or 6 N in case of N molecules in the asymmetric unit.
Examine the film to see how the rotation and translation steps relate the model
x1
(e.g.
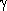 B Crystallin of which the structure was known)
and the target
x2
(e.g.
B Crystallin of which the structure was known)
and the target
x2
(e.g. 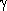 D Crystallin before that
its structure was known).
D Crystallin before that
its structure was known).
![]() Animated GIF "Movie" "Very High" Speed
Animated GIF "Movie" "Very High" Speed![]() Animated GIF "Movie" "High" Speed
Animated GIF "Movie" "High" Speed![]() Animated GIF "Movie" "Slow" Speed
Animated GIF "Movie" "Slow" Speed![]() Animated GIF "Movie" "Very Slow" Speed
Animated GIF "Movie" "Very Slow" Speed
![]() Welcome Page
Welcome Page
![]() Course Information
Course Information
![]() Course Index
Course Index
Copyright © 1995-2005 Birkbeck College
